Confusion With Acetaminophen
The Case
Parents brought their 5-year-old son to the emergency department (ED) with a 24-hour history of fever, cough, and frontal headache. Physical examination, vital signs, and laboratory evaluation were unremarkable. The patient was discharged with a diagnosis of viral syndrome after receiving one dose of acetaminophen in liquid form. Two days later, the patient returned to the ED with continuing fever and new rigors, vomiting, lethargy, and right upper quadrant abdominal pain. Laboratory evaluation indicated that acetaminophen levels and PT/INR were elevated.
Further discussion with the parents revealed that they misread the instructions about administering liquid acetaminophen. They gave multiple doses of 20 mL (48 mg/mL solution equaling 960 mg per dose) instead of the correct dose for their 20-kg child (6 mL = 288 mg). The patient was admitted to the hospital, given intravenous N-acetylcysteine, and his symptoms improved over the succeeding days. His acetaminophen levels declined, and he was safely discharged home without further events.
The Commentary
Available as an over-the-counter medication in the United States since 1960, acetaminophen is considered safe and effective for the management of fever and pain in children. Few serious adverse effects have been reported. A recent study evaluating more than 28,000 children treated with acetaminophen failed to show any increased risk of acute gastrointestinal bleeding, acute renal failure, or anaphylaxis.(1)
Despite this enviable safety record, acetaminophen-induced hepatotoxicity can be lethal when the medicine is taken in supratherapeutic doses. These include an intentional overdose with a single dose exceeding 140 mg/kg, unintentional overdose when a child is given multiple doses of acetaminophen that exceed manufacturer recommendations, or inadvertent overdose when a child receives acetaminophen in combination with cough/cold preparations.(2) The frequency of accidental acetaminophen overdoses causing clinically significant hepatotoxicity as described in this case is unknown. However, during a 10-year period at five California hospitals, 73 children (younger than 19 years) presented with acetaminophen hepatotoxicity, with 62 of 63 suicidal patients (three required orthotopic liver transplant) and 9 of 10 patients with accidental overdoses surviving.(3) Another study of acetaminophen hepatotoxicity in accidental overdose cases reported a wide range of implicated doses, but the most frequent causes involved administration of adult preparations to children (approximately half of cases), followed by inaccurate substitution of a higher-concentration preparation for a lower one.(4) Fifty-five percent of the patients died, and five underwent liver transplants, with four surviving.
Pharmacology of Acetaminophen Overdose
As with single, large overdoses of acetaminophen, hepatotoxicity from multiple supratherapeutic doses also results when the normal metabolic pathways in the liver are overwhelmed by the volume of drug, which leads to a series of molecular events ultimately causing cellular death.(5) The severity of liver injury is dependent upon the quantity of acetaminophen ingested, whether the P450 cytochrome system has been induced with drugs or alcohol, and potentially the nutritional state of the patient. Unlike acute large ingestions of acetaminophen with suicidal intent, in which a nomogram (6) helps to predict outcome and need for N-acetylcysteine treatment, the nomogram offers little value in predicting outcome with multiple supratherapeutic doses. However, identifying elevated acetaminophen concentrations in the blood relative to the last dose may reveal a risk for hepatotoxicity. Although N-acetylcysteine is effective in preventing serious liver injury with acute toxic ingestions, there is no proof of its benefit when multiple excessive doses are taken.(7) Once a patient is identified with hepatotoxicity after chronic overdosage, only supportive therapy is helpful and, if liver failure develops, liver transplantation can be lifesaving.
Safety Strategies
A number of safety interventions could prevent future acetaminophen overdoses, and these efforts should target individual providers and parents, medication labeling practices, and broader health care systems. First, the key to prevention begins with parental education. Physicians’ office personnel, pharmacists, and all health care providers play an important role in educating parents regarding the safe use of over-the-counter medications such as acetaminophen. Education should begin with instructions about acetaminophen safety, including dosing and forms of available preparations, as well as emphasizing to parents that “more is not better” with acetaminophen use. In addition, checking whether acetaminophen-containing cough/cold preparations are being inadvertently used with acetaminophen provides an important cautionary strategy. Finally, preparing illustrative handouts that depict when acetaminophen is a sole ingredient versus a combination (cough/cold) product to indicate the concentrations of acetaminophen in each would be beneficial.
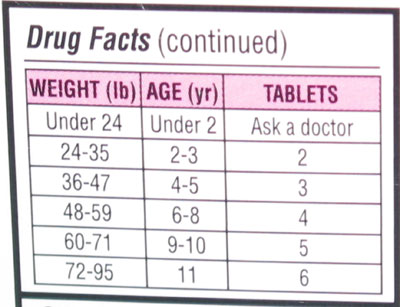
From a medication labeling perspective, encouraging manufacturers to limit liquid preparations to only a single concentration might prevent confusion regarding infant/child formulations. Currently, liquid acetaminophen comes in varying concentrations, thus creating potential for confusion (Figure 1), particularly for those parents with low health literacy. Perhaps this was the contributing factor in the case presented. In addition, highlighting (by bolding in the ingredients) the presence of acetaminophen in combination products might prevent unintended “double dosing.” Although acetaminophen labeling (Figure 2) provides guidelines on appropriate dosing, they are not weight based . But even weight-based labeling would not be a panacea, since it relies on accurate calculations that are more involved than current age-based dosing guidelines for children. In practice, the age-based guidelines should be sufficient to prevent the average-size child from receiving excessive dosing. Lastly, requiring pharmacies or drug manufacturers to prepare a “handout” (or display) at the point of sale with dosing instructions for parents might serve to emphasize safe administration practices.
To minimize acetaminophen drug toxicity, encourage weight-based dosing of 10-15 mg/kg/dose and do not exceed 5 doses in 24 hours. It is also important to be aware of agents that might amplify acetaminophen toxicity and adjust dosing accordingly using a pharmacist or a readily available drug reference resource for guidance (eg, PDA resources). Remind parents that acetaminophen is a safe and effective therapy but administering it in greater-than-recommended doses will not be more effective and may be harmful, even life-threatening.
Take-Home Points
- Acetaminophen hepatotoxicity after multiple supratherapeutic doses is a rare and preventable condition.
- Weight-based administration of acetaminophen is crucial in preventing supratherapeutic dosing.
- In treating infants or small children, select a single dose strength of acetaminophen and use it consistently.
- Always screen for concurrent use of cough/cold preparations containing acetaminophen if you plan to recommend acetaminophen for a child.
James E. Heubi, MD Professor of Pediatrics Assistant Dean for Clinical Research University of Cincinnati College of Medicine Program Director, General Clinical Research Center Cincinnati Children's Hospital Medical Center
References
1. Lesko SM, Mitchell AA. An assessment of the safety of pediatric ibuprofen. A practitioner-based randomized clinical trial. JAMA. 1995;273:929-933. [ go to PubMed ]
2. Heubi JE. Acetaminophen: the other side of the story. Contemp Pediatr. 1999;16:61-80.
3. Rivera-Penera T, Gugig R, Davis J, et al. Outcome of acetaminophen overdose in pediatric patients and factors contributing to hepatotoxicity. J Pediatr. 1997;130:300-304. [ go to PubMed ]
4. Heubi JE, Barbacci MB, Zimmerman HJ. Therapeutic misadventures with acetaminophen: hepatoxicity after multiple doses in children. J Pediatr. 1998;132:22-27. [ go to PubMed ]
5. Mitchell JR, Jollow DJ, Potter WZ, Gillette JR, Brodie BB. Acetaminophen-induced hepatic necrosis. IV. Protective role of glutathione. J Pharmacol Exp Ther. 1973;187:211-217. [ go to PubMed ]
6. Rumack BH, Matthew H. Acetaminophen poisoning and toxicity. Pediatrics. 1975;55:871-876. [ go to PubMed ]
7. Rumack BH. Acetaminophen overdose. Am J Med. 1983;75:104-112. [ go to PubMed ]
8. Heubi JE, Bien JP. Acetaminophen use in children: more is not better. J Pediatr. 1997;130:175-177. [ go to PubMed ]
Figures
Figure 1. Various options for over-the-counter children's acetaminophen. Product packaging emphasizes ages on these various formulations. Age ranges listed are different for the oral suspension (infant and 2-11) versus the rapidly dissolved tablets (2-6 and 6-11); the dosages in each tablet are higher for the older age range.
Figure 2. Table of dosage instructions on acetaminophen rapidly dissolved tablets (80 mg). Although the front of the package states that the tablets are for ages 2-6, the table on the back of the box (shown here) suggests dosing for children up to age 11.